| Gastroenterology Research, ISSN 1918-2805 print, 1918-2813 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Gastroenterol Res and Elmer Press Inc |
| Journal website https://gr.elmerpub.com |
Original Article
Volume 18, Number 1, February 2025, pages 23-30
Histone Lactylation-Driven Ubiquitin-Specific Protease 34 Promotes Cisplatin Resistance in Hepatocellular Carcinoma
Ming Fana, Jian Shan Liua, Xi Le Weia, Ye Niea, Hai Liang Liua, b
aDepartment of Hepatobiliary Surgery, The First Affiliated Hospital of Air Force Medical University, Xi’an 710032, Shaanxi, China
bCorresponding Author: Hai Liang Liu, Department of Hepatobiliary Surgery, The First Affiliated Hospital of Air Force Medical University, Xi’an 710032, Shaanxi, China
Manuscript submitted November 1, 2024, accepted December 24, 2024, published online January 17, 2025
Short title: USP34 Promotes Cisplatin Resistance in HCC
doi: https://doi.org/10.14740/gr1796
| Abstract | ▴Top |
Background: Ubiquitin-specific protease 34 (USP34) is a deubiquitinase that has been shown to play a critical role in the process of tumor drug-resistance. The objective of this study was to investigate the role of USP34 in cisplatin resistance in hepatocellular carcinoma (HCC).
Methods: Firstly, we analyzed the USP34 levels in cisplatin-sensitive and -resistant patients using The Cancer Genomic Atlas (TCGA) data from Gene Expression Profiling Interactive Analysis (GEPIA2). The cell viability and half-maximal inhibitory concentration (IC50) were measured by Cell Counting Kit-8 (CCK-8) assay. The cell apoptosis of HepG2 and HepG2/DDP cells was detected by annexin V-fluorescein isothiocyanate/propidium iodide (FITC/PI) double staining. The expression levels of USP34, multidrug resistance-associated protein 1 (MRP1), p-glycoprotein (p-gp), pan-lysine lactylation (Pan-Kla), histone H3 lysine 18 lactylation (H3K18la), lactate dehydrogenase A (LDHA) and lactate dehydrogenase B (LDHB) were measured by Western blot. HCC samples from the GEPIA2 database were used to determine the correlation between USP34 with LDHA and LDHB expression.
Results: USP34 was significantly upregulated in cisplatin-resistant HCC tissues and cells. Functional studies found that knockdown of USP34 inhibited HepG2 and HepG2/DDP cell proliferation and survival. Importantly, knockdown of USP34 enhanced cisplatin sensitivity in HepG2 and HepG2/DDP cells. Mechanistically, lactylation of histones promoted the expression level of USP34 in HepG2/DDP cells.
Conclusion: USP34 promotes the progression of HCC by regulating histone lactylation levels and cisplatin resistance in HCC.
Keywords: USP34; Lactylation; Hepatocellular carcinoma; Cisplatin
| Introduction | ▴Top |
Hepatocellular carcinoma (HCC) is a primary malignant tumor of the liver, commonly found in chronic liver disease and cirrhosis patients [1]. At present, various treatment strategies for HCC include surgical resection, chemotherapy, and liver transplantation [2]. Cisplatin is a commonly used chemotherapy drug in the clinical treatment of HCC, designed to kill cancer cells by inhibiting their DNA replication and division [3]. Although cisplatin has achieved certain therapeutic effects in the treatment of HCC, the emergence of drug-resistance has become a key factor limiting its therapeutic effect [4]. Therefore, a deep understanding of the cisplatin resistance mechanism in HCC is of great significance for improving treatment efficacy.
During cellular metabolism, the accumulation of lactic acid promotes the lactylation of histone lysine. Lactylation refers to the accumulation of lysine residues on lactylation modified histones during cellular metabolism [5]. As is well known, lactylation originates from the lactate produced by cellular glucose metabolism and is regulated by glycolysis and mitochondrial oxidative metabolism [6]. Previous studies have reported that lactylation is not only closely related to the energy metabolism of tumor cells, but also participates in regulating biological processes such as tumor cell growth, proliferation, and metastasis [7]. Especially the protein modification driven by lactate has been found to play an important role in the mechanism of tumor resistance [8]. These provide us with a new perspective to explore the mechanism of cisplatin resistance in HCC.
Ubiquitin-specific protease 34 (USP34) is a kind of deubiquitinase, which has been proved to be upregulated in many cancers, including pancreatic cancer [9], laryngeal squamous cell carcinoma [10], breast cancer [11], etc. In addition, USP34 is associated with cancer cell resistance to cisplatin. Research reports that knockdown of USP34 increases the drug sensitivity of laryngeal squamous cells to cisplatin and reduces cell survival rate [10]. As is well known, the main mechanism of action of cisplatin in the treatment of cancer is to form complexes with DNA, causing DNA strand breakage and damage, thereby interfering with the DNA synthesis and repair of cancer cells, inducing cell cycle arrest and apoptosis, and thereby inhibiting the proliferation and spread of cancer cells [12]. Interestingly, during DNA damage, USP34 stabilizes the RNF168 repair protein through deubiquitination, mediating DNA repair. In the absence of USP34, RNF168 is rapidly degraded, DNA repair is impaired, and cell survival rate decreases [13]. Therefore, we speculate that USP34 may be related to the mechanism of HCC cisplatin resistance.
The purpose of the research was to investigate the role of USP34 in cisplatin resistance and explore the relationship between the USP34 and histone lactylation in HCC. The results showed that interference with USP34 inhibited the proliferation and survival of HCC cells and increased the sensitivity of drug-resistant cells to cisplatin. The increased expression of USP34 in drug-resistant cells was associated with elevated levels of histone lactylation.
| Materials and Methods | ▴Top |
Tissue samples
Tumor tissues and adjacent normal tissues from 40 HCC patients at the hospital were collected, including 20 cisplatin-sensitive HCC patients and 20 cisplatin-resistant HCC patients. This study was reviewed and approved by the Ethics Committee of the First Affiliated Hospital of Air Force Medical University (No. KY20172013-1, date of approval: 2017-04-06). The study was performed in line with the principles of the Declaration of Helsinki.
Cell culture
Human normal liver cell line IHHA-1, HCC cell line HepG2 and cisplatin-resistant cell line HepG2/DDP were purchased from the American Type Culture Collection (ATCC, VA, USA). All cells were incubated with Dulbecco’s modified Eagle medium (DMEM) and 10% fetal bovine serum at 37 °C and 5% CO2.
Cell transfection
The interference RNA of USP34 (si-USP34-1, si-USP34-2), lactate dehydrogenase A (LDHA) (si-LDHA) and lactate dehydrogenase B (LDHB) (si-LDHB) were designed and synthesized by GenePharma (Shanghai, China). The respective siRNAs together with Lipofectamine 2000 (Invitrogen, CA, USA) were used for the cell transfection according to the manufacturer’s protocol. The cells were harvested for subsequent study following 48 h of transfection.
Real-time quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted from the sample and cells by the Trizol reagent (Invitrogen, CA, USA), and the concentration was determined. cDNA was synthesized using a reverse transcription kit (Takara, Shiga, Japan) according to the manufacturer’s protocol.
Cell viability analysis and half-maximal inhibitory concentration (IC50)
To evaluate the IC50 of HCC cells, cells were seeded in 96-well plates and tested via Cell Counting Kit-8 (CCK-8) assay. After culturing the cells overnight, 10 µL CCK-8 reagent was added and incubated in each well for further 4 h. The optical density (OD) value at 450 nm was evaluated by a microplate reader (Bio-Rad, CA, USA) to assess the cell viability. Finally, the IC50 of HCC cells for DDP was calculated.
Cell apoptosis
Apoptosis assays were performed using annexin V-fluorescein isothiocyanate (FITC) Apoptosis Assay Kit (Takara, Shiga, Japan) according to the manufacturer’s protocol. The treated cells were digested with trypsin, washed three times with phosphate-buffered saline (PBS) and then resuspended in 100 µL binding buffer. Subsequently, annexin-V and propidium iodide (PI) were added to the cell suspension and incubated for 15 min. Fluorescence intensity was detected by FACSCalibur flow cytometry (BD Biosciences, CA, USA) and apoptosis rate was calculated.
The Cancer Genomic Atlas (TCGA) dataset
In order to determine the correlation between USP34 and LDHA and LDHB expression in HCC, Gene Expression Profiling Interactive Analysis (GEPIA2) [14] was searched to obtain the transcription status of HCC samples.
Western blot
HCC cell lines were lysed using radioimmunoprecipitation assay (RIPA) lysate (Qiagen, NRW, Germany) and the levels of each protein were detected using the bicinchoninic acid (BCA) kit (Thermo Fisher Scientific, MA, USA). Equal proteins were isolated by 10% sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene fluoride (PVDF) membranes (Millipore, MA, USA). Following blocking for 2 h, the membranes were cultured with the indicated antibodies overnight at 4 °C. The primary antibodies were as follows: anti-USP34 (1:800), anti- multidrug resistance-associated protein 1 (MRP1) (1:800), anti-p-glycoprotein (p-gp) (1:800), anti-LDHA (1:800), anti-LDHB (1:800), anti-pan-lysine lactylation (Pan-Kla) (1:800), anti-histone H3 lysine 18 lactylation (H3K18la) (1:800) and anti-β-actin (1:2,000). After washing with Tris-buffered saline with Tween 20 (TBST) solution (Takara, Shiga, Japan), the membranes were incubated with secondary antibody (1:5,000) for 1 h, and the results were displayed using enhanced chemiluminescence (Pierce; Thermo Fisher Scientific, Inc.).
Statistical analysis
To ensure the reliability of the results, all experiments were performed with at least three sets of replications, and the findings were expressed as mean ± standard error of the mean (SEM). When comparing just two groups, t-test was used. However, for assessing disparities among multiple groups, one-way analysis of variance (ANOVA) was adopted followed by Tukey’s post hoc test.
| Results | ▴Top |
USP34 was increased in cisplatin-resistant HCC tissues and cells
USP34 levels in HCC and normal tissues were analyzed using TCGA data from GEPIA2, and it was found that USP34 was increased in HCC tumor tissue (Fig. 1a). Moreover, the mRNA of USP34 in tumor tissue was significantly upregulated, and the expression of USP34 in tumor tissue of cisplatin-resistant patients was increased than that of cisplatin-sensitive patients (Fig. 1b). In addition, HepG2 and HepG2/DDP cells were treated with different concentrations of cisplatin, and the cell viability of HepG2/DDP cells was promoted compared with HepG2 cells (Fig. 1c). It was found that the IC50 of HepG2/DDP cells was significantly higher compared with HepG2 cells (Fig. 1d). Finally, compared with IHHA-1 cells (normal human liver cells), USP34 mRNA and protein levels were elevated in HepG2 cells and further elevated in HepG2/DDP cells (Fig. 1e, f).
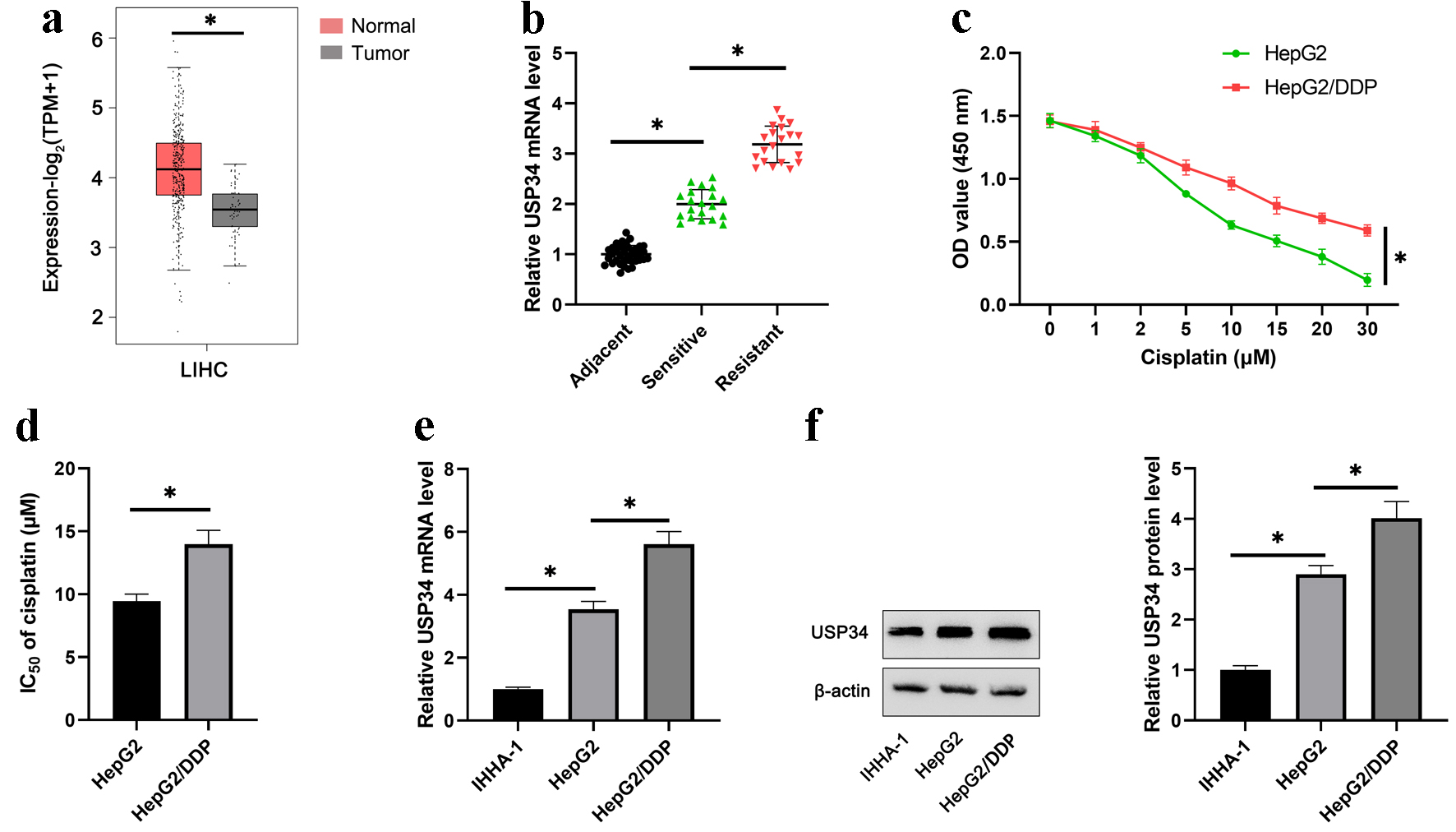 Click for large image | Figure 1. USP34 is significantly upregulated in cisplatin-resistant HCC tissues and cells. (a) The expression of USP34 in HCC and normal liver tissue using TCGA data from GEPIA2. (b) The expression of USP34 in tumor tissues of cisplatin-resistant patients and cisplatin-sensitive patients. (c) HepG2 and HepG2/DDP cells were treated with different concentrations (1, 2, 5, 10, 15, 20, 30 µM) of cisplatin for 48 h, and OD values were measured after 36 h. (d) The IC50 of HepG2 and HepG2/DDP cells. (e) The mRNA expression of USP34 in IHHA-1, HepG2 and HepG2/DDP cells. (f) The protein expression of USP34 in IHHA-1, HepG2 and HepG2/DDP cells. *P < 0.05. GEPIA2: Gene Expression Profiling Interactive Analysis; HCC: hepatocellular carcinoma; IC50: half-maximal inhibitory concentration; OD: optical density; TCGA: The Cancer Genomic Atlas; USP34: ubiquitin-specific protease 34. |
Knockdown of USP34 inhibited HepG2 and HepG2/DDP cell viability and survival
To explore the effect of USP34 on HCC, USP34 siRNAs (si-USP34-1, si-USP34-2) were transfected into HepG2 and HepG2/DDP cells. RT-qPCR and Western blot results showed a decrease in USP34 mRNA and protein levels, and the interference efficiency of si-USP34-2 was higher (Fig. 2a, b). So, subsequent experiments were conducted using si-USP34-2. Further research has found that interference with USP34 inhibited the cell viability (Fig. 2c) and promoted apoptosis of HepG2 and HepG2/DDP cells (Fig. 2d).
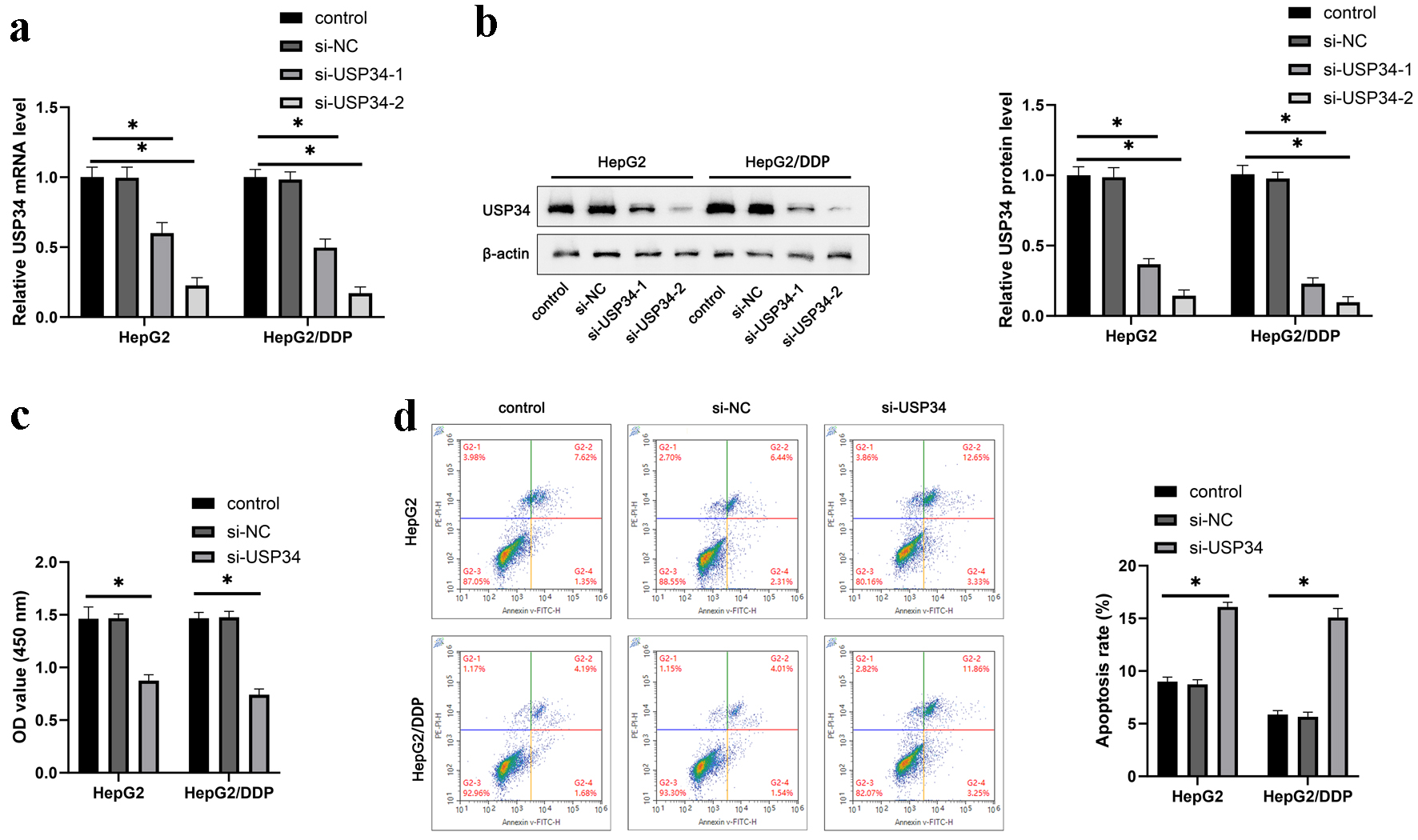 Click for large image | Figure 2. Knockdown of USP34 inhibits HepG2 and HepG2/DDP cell proliferation and survival. Constructing siRNA of USP34 (si-USP34-1, si-USP34-2) for transfection into HepG2 and HepG2/DDP cells. (a) The mRNA expression of USP34 in HepG2 and HepG2/DDP cells. (b) The protein expression of USP34 in HepG2 and HepG2/DDP cells. (c) The cell viability of HepG2 and HepG2/DDP cells. (d) The cell apoptosis of HepG2 and HepG2/DDP cells. *P < 0.05. USP34: ubiquitin-specific protease 34. |
Knockdown of USP34 enhanced cisplatin sensitivity in HepG2 and HepG2/DDP cells
To investigate the effect of USP34 on the sensitivity of HepG2 and HepG2/DDP cells to cisplatin, HepG2 and HepG2/DDP cells were treated with different concentrations of cisplatin after transfection with si-USP34. We found that knockdown of USP34 reduced the cell viability and IC50 of HCC cells (Fig. 3a-d). As expected, interference with USP34 significantly inhibited the protein levels of resistance-related proteins MRP1 and p-gp (Fig. 3e).
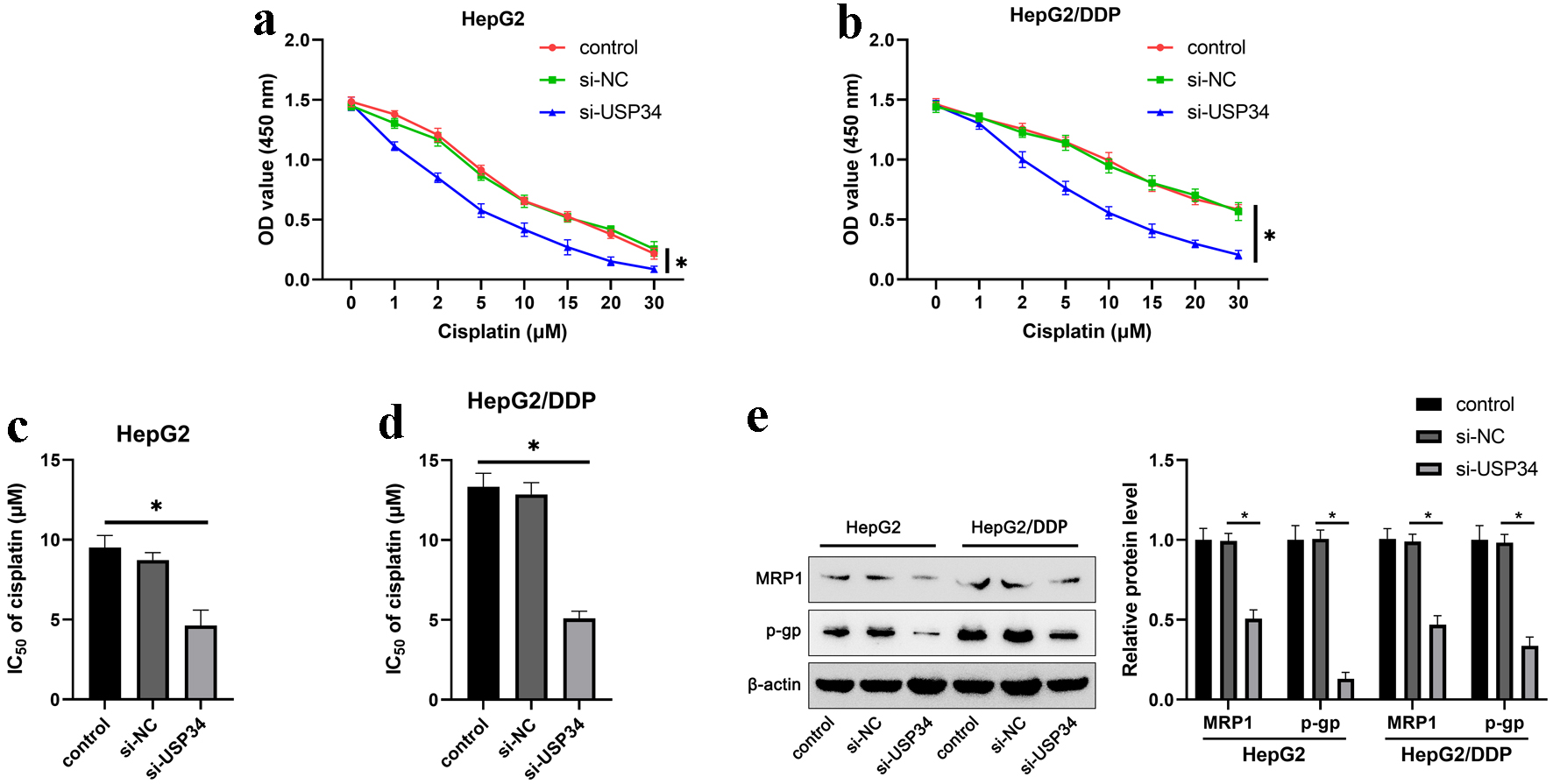 Click for large image | Figure 3. Knockdown of USP34 enhances cisplatin sensitivity in HepG2 and HepG2/DDP cells. HepG2 and HepG2/DDP cells were transfected with si-USP34 and treated with different concentrations (1, 2, 5, 10, 15, 20, 30 µM) of cisplatin. (a) The cell viability of HepG2. (b) The cell viability of HepG2/DDP cells. (c) The IC50 of HepG2 cells. (d) The IC50 of HepG2/DDP cells. (e) The protein expression of MRP1 and p-gp in HepG2 and HepG2/DDP cells. *P < 0.05. IC50: half-maximal inhibitory concentration; MRP1: multidrug resistance-associated protein 1; p-gp: p-glycoprotein; USP34: ubiquitin-specific protease 34. |
Upregulation of USP34 was associated with histone lactylation
Next, we investigated the potential mechanism of upregulation of USP34 in HCC. Firstly, we detected Pan-Kla and H3K18la, the most abundant form of histone lactylation, in IHHA-1, HepG2, and HepG2/DDP cells. The results revealed that the levels of Pan-Kla and H3K18la were upregulated in HepG2 cells, and further elevated in HepG2/DDP cells (Fig. 4a). The result suggests that a higher degree of lactylation may be related to the development and maintenance of drug-resistance.
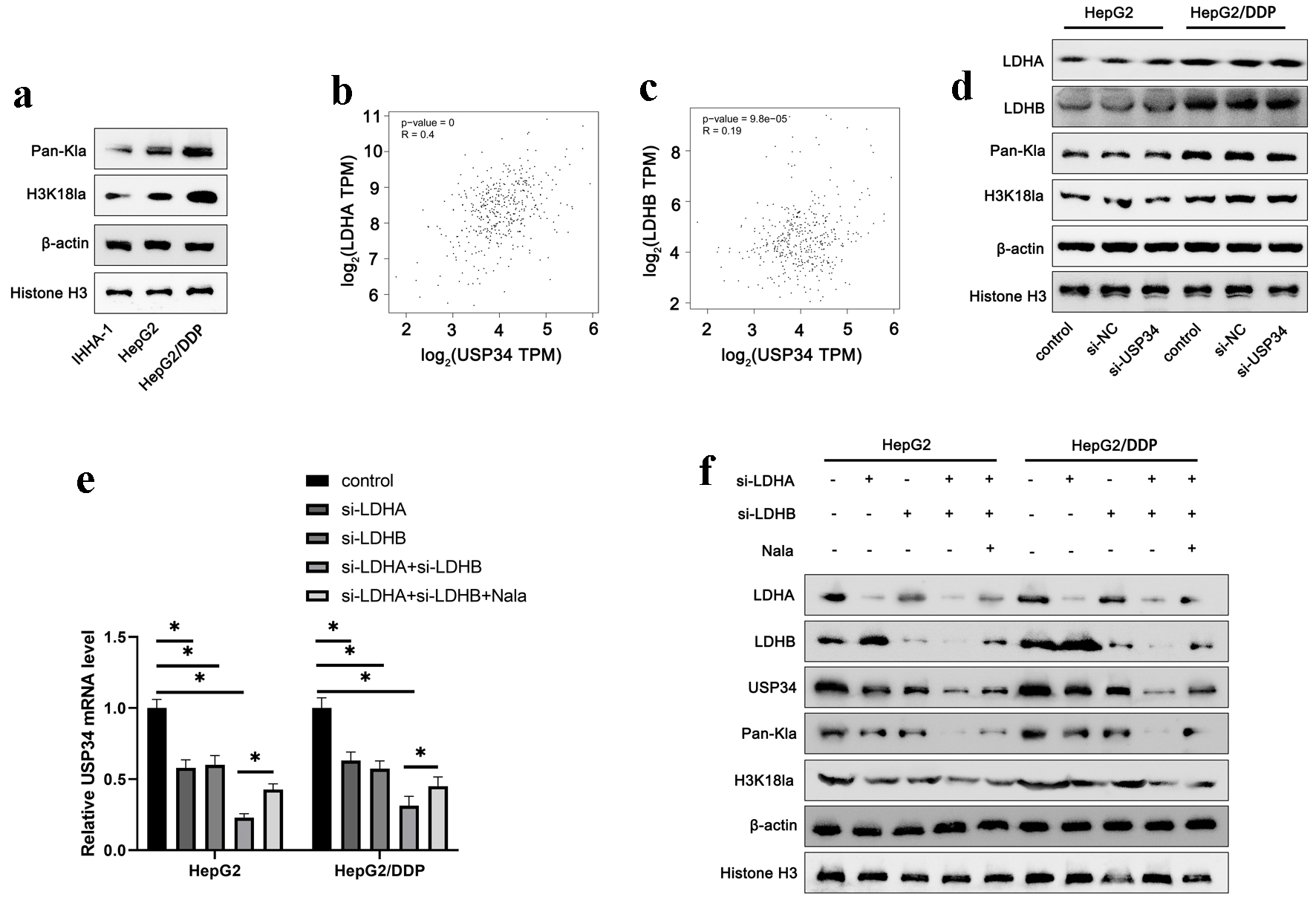 Click for large image | Figure 4. Upregulation of USP34 is associated with histone lactylation. (a) The protein expression of Pan-Kla and H3K18la in IHHA-1, HepG2 and HepG2/DDP cells. (b, c) The correlation between USP34 and LDHA and LDHB expression was analyzed using TCGA data from GEPIA2. Constructing siRNA of USP34 (si-USP34) for transfection into HepG2 and HepG2/DDP cells. (d) The levels of LDHA, LDHB, Pan-Kla and H3K18la in HepG2/DDP cells. HepG2 and HepG2/DDP cells were transfected with LDHA or LDHB siRNA (si-LDHA or si-LDHB) and then treated with sodium lactate (Nala). (e) The mRNA expression of USP34 in HepG2/DDP cells. (f) The levels of LDHA, LDHB, USP34, Pan-Kla and H3K18la in HepG2/DDP cells. *P < 0.05. GEPIA2: Gene Expression Profiling Interactive Analysis; H3K18la: histone H3 lysine 18 lactylation; LDHA: lactate dehydrogenase A; LDHB: lactate dehydrogenase B; Pan-Kla: pan-lysine lactylation; TCGA: The Cancer Genomic Atlas; USP34: ubiquitin-specific protease 34. |
Lactylation is closely related to lactate metabolism. Lactate dehydrogenase (LDH) plays a key role in the metabolism of lactic acid, responsible for catalyzing the production of lactic acid. Inhibiting LDH reduces histone lactylation, while exogenous addition of sodium lactate (Nala) increases the level of histone lactylation [6]. By analyzing TCGA data on GEPIA2, the results revealed that the expression of USP34 was positively correlated with LDHA and LDHB expression (Fig. 4b, c). In addition, we analyzed the correlation between USP34 and genes related to lactate production (PDHA1, PFKP, PKM) and lactate transport (SLC16A1) in HCC tissues, and found a significant positive correlation between USP34 and these genes (Supplementary Material 1, gr.elmerpub.com). Interference with USP34 did not affect the levels of LDHA, LDHB, Pan-Kla, and H3K18la (Fig. 4d). On the contrary, silencing of LDHA and LDHB decreased the mRNA and protein levels of USP34 (Fig. 4e, f), as well as Pan-Kla and H3K18la levels (Fig. 4f). In addition, exogenous supplementation of 15 µM Nala partially reversed the effects of silencing of LDHA and LDHB (Fig. 4e, f). This suggests that the upregulation of USP34 may be related to histone lactylation.
| Discussion | ▴Top |
USP34 is an important member of the deubiquitinase family and plays a key regulatory role in the progression of many tumors [15]. Research reports that USP34 stabilizes the expression of Pin1 through deubiquitylation and promotes Ubc9 isomerization in glioma stem cells, maintaining the stemness of glioma stem cells and promoting the growth of glioblastoma [16]. In addition, the level of USP34 was upregulated in pancreatic cancer, and USP34 promoted the proliferation and migration of pancreatic cancer cells, as well as inhibited cell apoptosis [17]. In addition, Dai et al reported that USP34 was increased in cisplatin-resistant laryngeal squamous cell carcinoma cells, and knockdown of USP34 increased the drug sensitivity of resistant cells to cisplatin, indicating a close correlation between USP34 and cisplatin resistance [10]. As is well known, changes in tumor lesion size are closely related to the chemotherapy drug-resistance [18]. The mechanism of cisplatin treatment for tumors is to disrupt the structure and function of tumor cell DNA, interfere with tumor cell DNA repair, and thereby inhibit tumor cell proliferation. The role of USP34 in mesenchymal stem cells and DNA damage repair has been reported [13, 19]. Therefore, we believe that USP34 may be related to the mechanism of HCC cisplatin resistance. This study found that the expression levels of USP34 were increased in cisplatin-resistant HCC tissues and cells, and interference with USP34 increased cisplatin sensitivity in HCC cells.
Histone lactylation is a novel post-translational modification of proteins, which is related to cellular metabolism and gene expression regulation, and has attracted much attention in tumor biology [20]. Lactylation is widely involved in the metabolic regulation process of the liver, which includes non-liver specific metabolic pathways such as sugar, protein and lipid metabolism, as well as liver specific metabolic pathways such as bile acid metabolism and drug metabolism [21, 22]. Research has found that the overall trend of lactylation is higher in cancer tissues than in adjacent tissues in HCC, indicating a potential connection between lactylation modification and the occurrence and development of HCC [23]. In addition, oncogenic M2 macrophages isolated from melanoma and lung cancer tissues in mice were significantly positively correlated with lactylation levels, suggesting that high lactylation and histone lactylation levels in macrophages may contribute to tumor formation and malignant progression [24, 25]. It is worth noting that there are reports that lactylation is closely related to drug-resistance in cancer cells. Li et al reported that lactylation drives cisplatin resistance in bladder cancer and accelerates cancer progression [26]. Consistent with this, our research found that the level of lactylation increased in cisplatin-resistant HCC cells, suggesting that a higher degree of lactylation is associated with the development and maintenance of drug-resistance.
Lactylation is a method of modifying proteins by introducing lactic acid groups (Lysine) into the protein molecule. Lactylation is widely present in living organisms and has a significant impact on the function and stability of proteins [27]. It can regulate the cellular localization, interaction, stability, and activity of proteins [28]. With the rapid development of proteomics technology, the application of lactylation in proteomics research is receiving increasing attention [29]. Chen et al reported that lactate promotes macrophage polarization towards M2-like phenotype through the lactylation of histone lysine, thereby inhibiting the immune response within the tumor microenvironment and exerting anti-cancer effects [30]. Twenty-eight lysine lactylation (Kla) sites were identified on core histones such as H3, H4, H2A and H2B in human HeLa cells and mouse bone marrow-derived macrophages (BMDMs) [31]. Four target genes regulated by H3K18 lactylation modification were found in melanoma cell lines. Among them, YTHDF2 was highly expressed in the tissues of melanoma patients, and the higher the expression level of YTHDF2, the worse the prognosis indicators of patients. This indicates that H3K18 lactylation modification affects the occurrence and development of melanoma by regulating the expression of YTHDF2 [6]. The above results suggest that lactylation modification is involved in the progression of various tumors by regulating the expression of key proteins.
In summary, the results of this study showed that USP34 was upregulated in HCC tissue, and its expression was higher in HCC tissue of cisplatin-resistant patients. In addition, interference with USP34 inhibited the proliferation and survival of HCC cells, and increased the sensitivity of drug-resistant cells to cisplatin. Mechanism research has found that histone lactylation induced increased expression of USP34, which was involved in the progression and regulation of HCC. It suggests that histone lactylation mediated USP34 plays a critical regulatory role in the cisplatin resistance of HCC (Fig. 5). Although this is a gratifying result, there are still some shortcomings in the current research. In the future, we need further research to evaluate off target effects and their roles in non-cancer cells to ensure clinical safety.
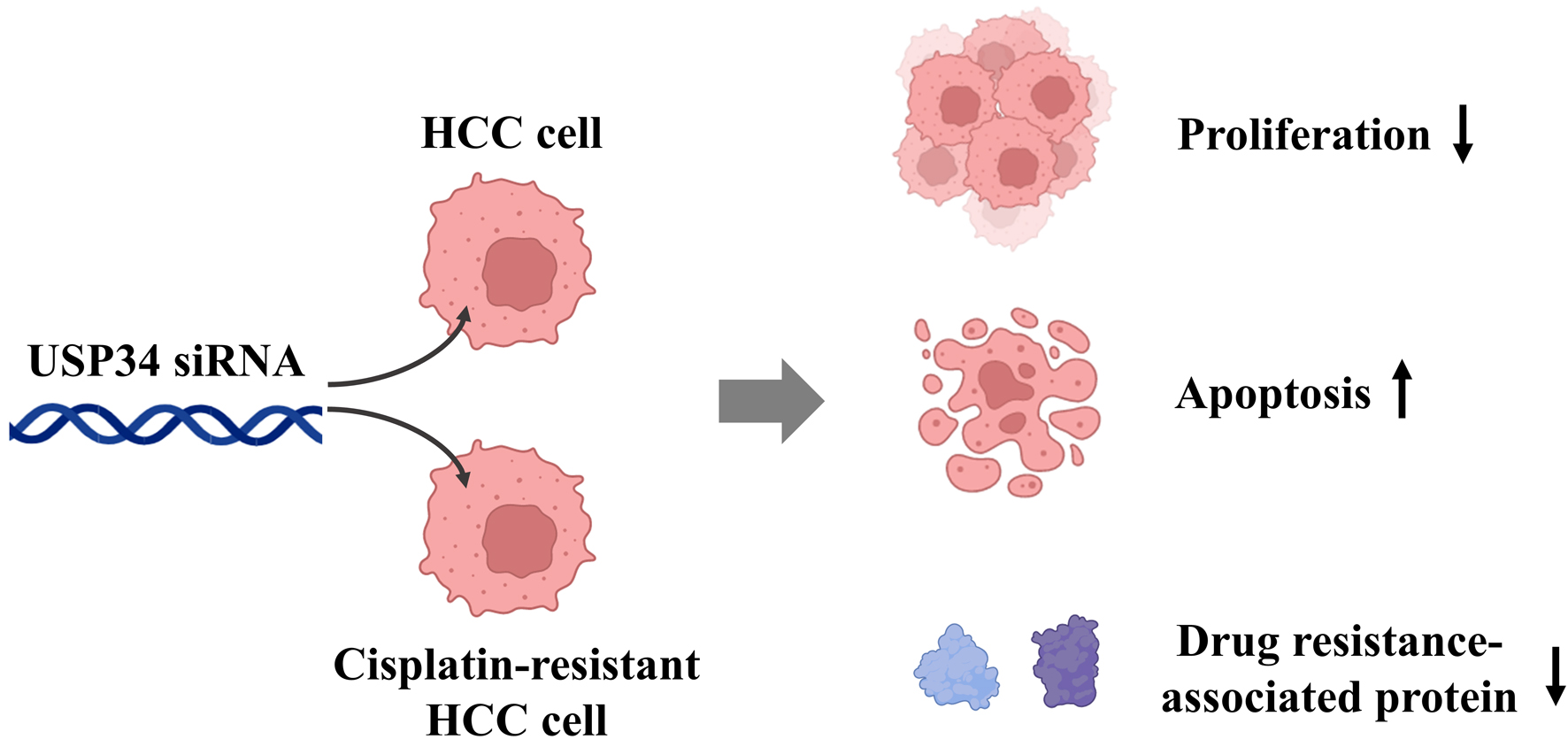 Click for large image | Figure 5. In HCC cells or cisplatin-resistant HCC cells, interference with USP34 inhibited cell proliferation, promoted cell apoptosis, and enhanced cisplatin sensitivity. HCC: hepatocellular carcinoma; USP34: ubiquitin-specific protease 34. |
| Supplementary Material | ▴Top |
Suppl 1. The expression of USP34 is positively correlated with PDHA1, PFKP, PKM and SLC16A1 expression in HCC tissues. (A-D) The correlation between USP34 and PDHA1, PFKP, PKM and SLC16A1 expression was analyzed using TCGA data from GEPIA2.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
All authors have completed the ICMJE uniform disclosure form. The authors have no conflict of interest to declare.
Informed Consent
Written informed consent was obtained from individual or guardian participants.
Author Contributions
Hai Liang Liu contributed to the study conception and design. Xi Le Wei and Ye Nie contributed to the material preparation and data collection. Jian Shan Liu contributed to data analysis and software. The first draft of the manuscript was written by Ming Fan. All authors commented on previous versions of the manuscript, read and approved the final manuscript.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
| References | ▴Top |
- Ganesan P, Kulik LM. Hepatocellular carcinoma: new developments. Clin Liver Dis. 2023;27(1):85-102.
doi pubmed - Gilles H, Garbutt T, Landrum J. Hepatocellular carcinoma. Crit Care Nurs Clin North Am. 2022;34(3):289-301.
doi pubmed - Hamaya S, Fujihara S, Iwama H, Fujita K, Shi T, Nakabayashi R, Mizuo T, et al. Characterization of cisplatin effects in lenvatinib-resistant hepatocellular carcinoma cells. Anticancer Res. 2022;42(3):1263-1275.
doi pubmed - Hamaya S, Oura K, Morishita A, Masaki T. Cisplatin in liver cancer therapy. Int J Mol Sci. 2023;24(13):10858.
doi pubmed - Lv X, Lv Y, Dai X. Lactate, histone lactylation and cancer hallmarks. Expert Rev Mol Med. 2023;25:e7.
doi pubmed - Yu J, Chai P, Xie M, Ge S, Ruan J, Fan X, Jia R. Histone lactylation drives oncogenesis by facilitating m(6)A reader protein YTHDF2 expression in ocular melanoma. Genome Biol. 2021;22(1):85.
doi pubmed - Qu J, Li P, Sun Z. Histone lactylation regulates cancer progression by reshaping the tumor microenvironment. Front Immunol. 2023;14:1284344.
doi pubmed - Li W, Zhou C, Yu L, Hou Z, Liu H, Kong L, Xu Y, et al. Tumor-derived lactate promotes resistance to bevacizumab treatment by facilitating autophagy enhancer protein RUBCNL expression through histone H3 lysine 18 lactylation (H3K18la) in colorectal cancer. Autophagy. 2024;20(1):114-130.
doi pubmed - Lin C, Xia J, Gu Z, Meng Y, Gao D, Wei S. Downregulation of USP34 inhibits the growth and migration of pancreatic cancer cells via inhibiting the PRR11. Onco Targets Ther. 2020;13:1471-1480.
doi pubmed - Dai WL, Yuan SX, Cao JP. The deubiquitinase USP34 stabilizes SOX2 and induces cell survival and drug resistance in laryngeal squamous cell carcinoma. Kaohsiung J Med Sci. 2020;36(12):983-989.
doi pubmed - Oh E, Kim JY, Sung D, Cho Y, Lee N, An H, Kim YJ, et al. Inhibition of ubiquitin-specific protease 34 (USP34) induces epithelial-mesenchymal transition and promotes stemness in mammary epithelial cells. Cell Signal. 2017;36:230-239.
doi pubmed - Dasari S, Tchounwou PB. Cisplatin in cancer therapy: molecular mechanisms of action. Eur J Pharmacol. 2014;740:364-378.
doi pubmed - Sy SM, Jiang J, O WS, Deng Y, Huen MS. The ubiquitin specific protease USP34 promotes ubiquitin signaling at DNA double-strand breaks. Nucleic Acids Res. 2013;41(18):8572-8580.
doi pubmed - http://gepia2.cancer-pku.cn.
- Zhao L, Tang Y, Yang J, Lin F, Liu X, Zhang Y, Chen J. Integrative analysis of circadian clock with prognostic and immunological biomarker identification in ovarian cancer. Front Mol Biosci. 2023;10:1208132.
doi pubmed - Zhu Q, Liang P, Meng H, Li F, Miao W, Chu C, Wang W, et al. Stabilization of Pin1 by USP34 promotes Ubc9 isomerization and protein sumoylation in glioma stem cells. Nat Commun. 2024;15(1):40.
doi pubmed - Gu Z, Lin C, Hu J, Xia J, Wei S, Gao D. USP34 regulated human pancreatic cancer cell survival via AKT and PKC pathways. Biol Pharm Bull. 2019;42(4):573-579.
doi pubmed - Blagosklonny MV. My battle with cancer. Part 1. Oncoscience. 2024;11:1-14.
doi pubmed - Guo YC, Wang MY, Zhang SW, Wu YS, Zhou CC, Zheng RX, Shao B, et al. Ubiquitin-specific protease USP34 controls osteogenic differentiation and bone formation by regulating BMP2 signaling. EMBO J. 2018;37(20):e99398.
doi pubmed - Sun L, Zhang Y, Yang B, Sun S, Zhang P, Luo Z, Feng T, et al. Lactylation of METTL16 promotes cuproptosis via m(6)A-modification on FDX1 mRNA in gastric cancer. Nat Commun. 2023;14(1):6523.
doi pubmed - Cheng Z, Huang H, Li M, Liang X, Tan Y, Chen Y. Lactylation-related gene signature effectively predicts prognosis and treatment responsiveness in hepatocellular carcinoma. Pharmaceuticals (Basel). 2023;16(5):644.
doi pubmed - Xu H, Li L, Wang S, Wang Z, Qu L, Wang C, Xu K. Royal jelly acid suppresses hepatocellular carcinoma tumorigenicity by inhibiting H3 histone lactylation at H3K9la and H3K14la sites. Phytomedicine. 2023;118:154940.
doi pubmed - Yang Z, Yan C, Ma J, Peng P, Ren X, Cai S, Shen X, et al. Lactylome analysis suggests lactylation-dependent mechanisms of metabolic adaptation in hepatocellular carcinoma. Nat Metab. 2023;5(1):61-79.
doi pubmed - Zhu Y, Song B, Yang Z, Peng Y, Cui Z, Chen L, Song B. Integrative lactylation and tumor microenvironment signature as prognostic and therapeutic biomarkers in skin cutaneous melanoma. J Cancer Res Clin Oncol. 2023;149(20):17897-17919.
doi pubmed - Jiang J, Huang D, Jiang Y, Hou J, Tian M, Li J, et al. Lactate modulates cellular metabolism through histone lactylation-mediated gene expression in non-small cell lung cancer. Front Oncol. 2021;11:647559.
- Li F, Zhang H, Huang Y, Li D, Zheng Z, Xie K, Cao C, et al. Single-cell transcriptome analysis reveals the association between histone lactylation and cisplatin resistance in bladder cancer. Drug Resist Updat. 2024;73:101059.
doi pubmed - Fan H, Yang F, Xiao Z, Luo H, Chen H, Chen Z, Liu Q, et al. Lactylation: novel epigenetic regulatory and therapeutic opportunities. Am J Physiol Endocrinol Metab. 2023;324(4):E330-E338.
doi pubmed - Zhang D, Tang Z, Huang H, Zhou G, Cui C, Weng Y, Liu W, et al. Metabolic regulation of gene expression by histone lactylation. Nature. 2019;574(7779):575-580.
doi pubmed - Zhang N, Zhang Y, Xu J, Wang P, Wu B, Lu S, Lu X, et al. alpha-myosin heavy chain lactylation maintains sarcomeric structure and function and alleviates the development of heart failure. Cell Res. 2023;33(9):679-698.
doi pubmed - Chen L, Huang L, Gu Y, Cang W, Sun P, Xiang Y. Lactate-lactylation hands between metabolic reprogramming and immunosuppression. Int J Mol Sci. 2022;23(19):11943.
doi pubmed - Sun L, Zhang H, Gao P. Metabolic reprogramming and epigenetic modifications on the path to cancer. Protein Cell. 2022;13(12):877-919.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Gastroenterology Research is published by Elmer Press Inc.