| Gastroenterology Research, ISSN 1918-2805 print, 1918-2813 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Gastroenterol Res and Elmer Press Inc |
| Journal website https://gr.elmerpub.com |
Original Article
Volume 19, Number 2, April 2026, pages 64-73
Evaluating Glucagon-Like Peptide-1 Receptor Agonist Safety Before Upper Endoscopy: A Systematic Review and Meta-Analysis
Zohaib Ahmeda, k, Amna Iqbala, Sami Ghazaleha, Abdallah Kobeissya, Ali Nawrasa, Yaseen Alastala, Hani Saadb, Sarmed Mansurb, Idrees Mohiuddinb, Umair Iqbalc, Dushyant Singh Dahiyad, Syeda Faiza Arife, Connor Campbellf, Wade Lee-Smithg, Mohammad W. Asifh, Faisal Kamali, Aamir Saeedj, Omar Horanib
aDivision of Gastroenterology and Hepatology, University of Toledo College of Medicine and Life Sciences, Toledo, OH, USA
bDivision of Hospital Medicine, University of Toledo College of Medicine and Life Sciences, Toledo, OH, USA
cDivision of Gastroenterology and Hepatology, WellSpan Health, York, PA, USA
dDivision of Gastroenterology and Hepatology, University of Kansas, Lawrence, KS, USA
eDow University of Health Sciences, Karachi, Pakistan
fUniversity of Toledo Medical Science Health Campus, Toledo, OH, USA
gUniversity Libraries, University of Toledo, Toledo, OH, USA
hDepartment of Neuroscience, Ohio State University, Columbus, OH, USA
iJefferson Health-Thomas Jefferson University Hospitals, Philadelphia, PA, USA
jVanderbilt University Medical Center, Nashville, TN, USA
kCorresponding Author: Zohaib Ahmed, Division of Gastroenterology, The University of Toledo College of Medicine and Life Sciences, Toledo, OH 43614, USA
Manuscript submitted January 27, 2026, accepted April 9, 2026, published online April 27, 2026
Short title: GLP-1 RA Safety in Upper Endoscopy
doi: https://doi.org/10.14740/gr2108
| Abstract | ▴Top |
Background: The use of glucagon-like peptide-1 receptor agonists (GLP-1 RAs) has remarkably increased in the past few years as they have demonstrated significant reduction in A1c and major cardiovascular outcomes and have led to weight loss with low risk of hypoglycemia. GLP-1 RA use has raised concerns about potential associations with increased gastric retention and adverse outcomes in upper endoscopy procedures.
Methods: We conducted a comprehensive search across multiple databases to identify comparative studies evaluating the impact of GLP-1 RA exposure versus non-exposure on upper endoscopy outcomes. Studies comparing GLP-1 RAs to other medications were excluded. The primary outcome was the presence of retained gastric contents, with secondary outcomes including aspiration risk and the need for repeated esophagogastroduodenoscopy (EGD).
Results: Our systematic review included 28 studies, comprising data from 212,082 individuals collected between 2015 and 2023. The meta-analysis revealed that GLP-1 RA use was associated with approximately four times higher odds of retained gastric contents compared to non-exposure (odds ratio (OR): 3.95, confidence interval (CI): 2.691–5.815, P < 0.0001, I2: 82.71%). There were no increased odds of aspiration (OR: 1.09, CI: 0.459–2.600, P = 0.145, I2: 91.44%) or repeated upper endoscopies (OR: 1.82, CI: 0.813–4.085, P = 0.145, I2: 85.32%). However, GLP-1 RA use was significantly associated with increased odds of procedure cancellation (OR: 3.96, CI: 2.870–5.563, P < 0.0001, I2: 4.19%).
Conclusion: While GLP-1 RAs do not increase the risk of aspiration or necessitate repeated procedures, they are linked to a higher incidence of retained gastric contents and procedure cancellations. Further randomized controlled trials are necessary to better understand the effects of GLP-1 RAs on upper endoscopy outcomes.
Keywords: Glucagon-like peptide-1 receptor agonists; Retained gastric contents; Upper endoscopy; EGD
| Introduction | ▴Top |
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are widely used for management of type 2 diabetes mellitus (T2DM) and obesity due to their effectiveness in regulating serum blood glucose levels and promoting weight loss [1]. Common side effects of GLP-1 RAs include nausea, vomiting, diarrhea, and constipation [2]. Given their cardiometabolic benefit [3, 4] and weight loss effects [5], their global usage has increased from 1.2% in 2014 to 15% in 2022 in patients with type 2 diabetes mellitus and cardiovascular risk factors [6, 7].
GLP-1 RA use prior to upper endoscopy has raised concerns regarding the safety of these medications [8–10]. According to available data, aspiration and unintentional intubations pose a potential hazard due to risk of aspiration pneumonia and high rates of in-hospital mortality [11, 12]. Moreover, retained gastric contents (RGCs) and the need for repeat endoscopy are significant concerns, as they can impair procedure visibility and lead to unnecessary utilization of healthcare resources [13].
With the rise of GLP-1 RA use, the American Society of Anesthesiologists released a statement [14] advising consideration of holding GLP-1 RA prior to endoscopy, which prompted a multi-gastroenterology society statement [15, 16] concluding that there were no actionable data to suggest stopping GLP-1 RA prior to endoscopy. This illustrated competing concerns regarding GLP-1 RA use prior to endoscopy. Several more recent studies have emerged [17–19] identifying a correlation between gastric residue on esophagogastroduodenoscopy (EGD) and GLP-1 RA use.
Despite the rapid expansion of GLP-1 RA use and growing concerns regarding delayed gastric emptying, there remains a lack of high-quality, consolidated evidence evaluating their impact on clinically relevant endoscopic outcomes, including RGCs, aspiration risk, and procedure cancellation. This knowledge gap contributes to ongoing uncertainty in peri-procedural management and inconsistent recommendations regarding whether these agents should be withheld prior to endoscopy. This study aims to systematically review the literature and perform a meta-analysis to evaluate the association between GLP-1 RA use and endoscopic outcomes, including RGCs, aspiration events, procedure cancellation, and repeat endoscopy.
| Materials and Methods | ▴Top |
Search strategy
A comprehensive search strategy to identify studies reporting the effects of GLP-1 agonists on stomach emptying or mucosal visibility in EGD procedures was constructed in Embase (Embase.com, Elsevier) by an experienced health sciences librarian (WL-S) using truncated keywords, phrases, and subject headings. This strategy was translated to MEDLINE (PubMed platform, National Center for Biotechnology Information, National Library of Medicine), Cochrane Central Register of Controlled Trials (CochraneLibrary.com, Wiley), and the Web of Science Core Collection (Web of Science platform, Clarivate) with all searches performed on June 25, 2024 (Supplementary Material 1, gr.elmerpub.com). No publication date or language limits were used. All results were exported to EndNote 21 citation management software (Clarivate, Philadelphia, PA, USA) and duplicates were removed by successive iterations of EndNote’s duplicate detection algorithms and manual inspection. We adhered to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [20] (Supplementary Material 2, gr.elmerpub.com). No language restrictions were applied.
Inclusion and exclusion criteria
Two study authors (ZA and SD) reviewed records. Animal studies, pediatric populations, review articles, case reports, and case series were excluded. Studies that did not assess outcomes related to upper endoscopy, single arm studies without a control group, and studies comparing GLP-1 RAs to other medications were also excluded. Studies that met the following criteria included randomized controlled trials and prospective or retrospective cohort studies that compared endoscopy outcomes with or without prior exposure to GLP 1-RA, and reported outcomes of interest. Abstracts were included given limited availability of literature on this topic prior to 2022 to minimize publication bias [21]. Inclusion of each study was agreed upon by two authors (ZA and SD) and a third reviewer (AI) resolved any discrepancies in study inclusion.
Data extraction and study outcomes
The following data were extracted from the studies: author name, publication year, country of origin, study design, gender and age of patients, and total upper endoscopy procedures (Supplementary Material 3, gr.elmerpub.com). Outcome measures were retrieved with RGCs as the primary outcome. Most studies reported an adjusted odds ratio (OR), which was recorded; otherwise, the number of patients with RGC over total events was recorded. Secondary outcomes included aspiration events (aspiration, aspiration pneumonia, and aspiration pneumonitis), as reported by individual studies. Due to inconsistent and non-uniform reporting, these outcomes were pooled as a composite “aspiration events” outcome. Other outcomes included number of repeat endoscopy procedures and procedure cancellations, which were extracted and recorded as events of interest over total number of procedures (Supplementary Material 4, gr.elmerpub.com). Of note, events of interest and total number of procedures from propensity-matched comparison groups were recorded to limit confounders. Data were collected and tabulated on Microsoft Excel (Microsoft, USA) by two independent reviewers (AI and SD). Any discrepancy in data collection was resolved through mutual discussion with a third reviewer (ZA). Definitions of RGCs were not fully standardized across included studies. In most studies, RGC was defined as the presence of visible food residue in the stomach at the time of upper endoscopy despite adherence to standard fasting protocols. However, some studies categorized RGC qualitatively (e.g., minimal residue vs. significant retained contents) or based on procedural impact (e.g., impaired visualization or procedure interruption). This variability in outcome definition was considered during data interpretation. When available, information regarding timing of the last GLP-1 RA dose prior to endoscopy was extracted; however, this was inconsistently reported across studies. When available, information regarding timing of GLP-1 RA use prior to endoscopy was extracted; however, this was inconsistently reported across studies. For studies reporting both unmatched and propensity-matched cohorts, data from the propensity-matched cohorts were preferentially extracted and included in the meta-analysis to minimize confounding. Unmatched cohorts from the same studies were not included to avoid duplication.
Data synthesis and analytical analysis
Comprehensive Meta-Analysis (CMA) Software Version 4 (Bio STAT, Englewood, New Jersey, USA) was used to perform meta-analysis. For the primary outcome of RGC, reported adjusted ORs and CIs were entered as reported by the study; otherwise the ORs and CIs were calculated from events of interest over the total number of procedures. ORs and CIs were pooled to determine an overall effect size. Several included studies reported adjusted ORs while others only provided event data from which crude ORs were calculated. To maximize inclusion of available evidence, both adjusted and calculated crude ORs were pooled using a random-effects model. For secondary outcomes, events of interest over the total number of procedures were used to calculate ORs and CIs that were pooled to determine an overall effect size. The random effects model was used with a confidence level of 95%. Statistical significance was defined as a P-value of < 0.05. Heterogeneity was assessed using the Higgins I2 index, where I2 values > 50% implied the presence of substantial heterogeneity [22].
For outcomes with 10 or more studies, a funnel plot was generated, and visual symmetry was assessed to comment on publication bias for each outcome (Supplementary Material 5, gr.elmerpub.com).
For the primary outcome, data from greater than three peer-reviewed manuscripts were available, so we conducted sensitivity analysis for RGC excluding abstracts (Supplementary Material 6a, gr.elmerpub.com). Additionally, for outcomes with 10 or more studies including RGC and cancelled procedures, sensitivity analysis was performed excluding each study individually to assess the resultant effect size (Supplementary Material 6b, c, gr.elmerpub.com). Sensitivity analysis was performed to assess the robustness of pooled effects. No subgroup analyses were performed compared to individual GLP-1 RAs as most studies assessed a mixture of GLP-1 RA agents compared to a control.
Bias assessment
The bias assessment for included studies was evaluated using Newcastle-Ottawa scale for observational studies [23]. Publication bias was visually assessed using funnel plots and quantitatively assessed using Egger’s regression analysis. A P-value < 0.05 was indicative of substantial publication bias. Publication bias was visually assessed using funnel plots and quantitatively assessed using Egger’s regression analysis. A P-value < 0.05 was indicative of substantial publication bias.
| Results | ▴Top |
Study selection
The search strategy produced 145 articles, 49 of which were removed as duplicates (PRISMA flow chart, Fig. 1). Of the remaining 96 articles, 61 were removed after title and abstract review. No additional relevant articles were identified from a review of bibliographies. After the full texts of the remaining 35 articles were reviewed, we excluded two non-comparative studies. Three studies were excluded as they did not include relevant data. Two studies were excluded as they were case reports. A total of 28 retrospective studies [8, 10, 24–48] including 21 cohort studies, six case-control studies, and one cross-sectional study were included in final analysis.
 Click for large image | Figure 1. PRISMA 2020 flow diagram for systematic review if databases, registers, and other sources. |
Study characteristics
Our final analysis included 212,082 patients with an average mean reported age of 56.2 years, 60.1% of the population were female, 56.3% had T2DM, and mean BMI was 34.2. Four studies assessed semaglutide alone with the remaining studies reporting a mixed group of GLP-1 RAs. All studies included patients who had GLP-1 exposure of at least 3–4 months.
Meta-analysis
Primary outcome
Our pooled analysis revealed a higher odd of RGC in the GLP-1 RA group compared to the group with no prior exposure to GLP-1 RA (OR: 3.95, CI: 2.691–5.815, P < 0.0001, I2: 82.71%) (Fig. 2). Across included studies, the reported incidence of retained gastric contents varied, with higher rates observed in patients receiving GLP-1 RAs compared to non-exposed patients.
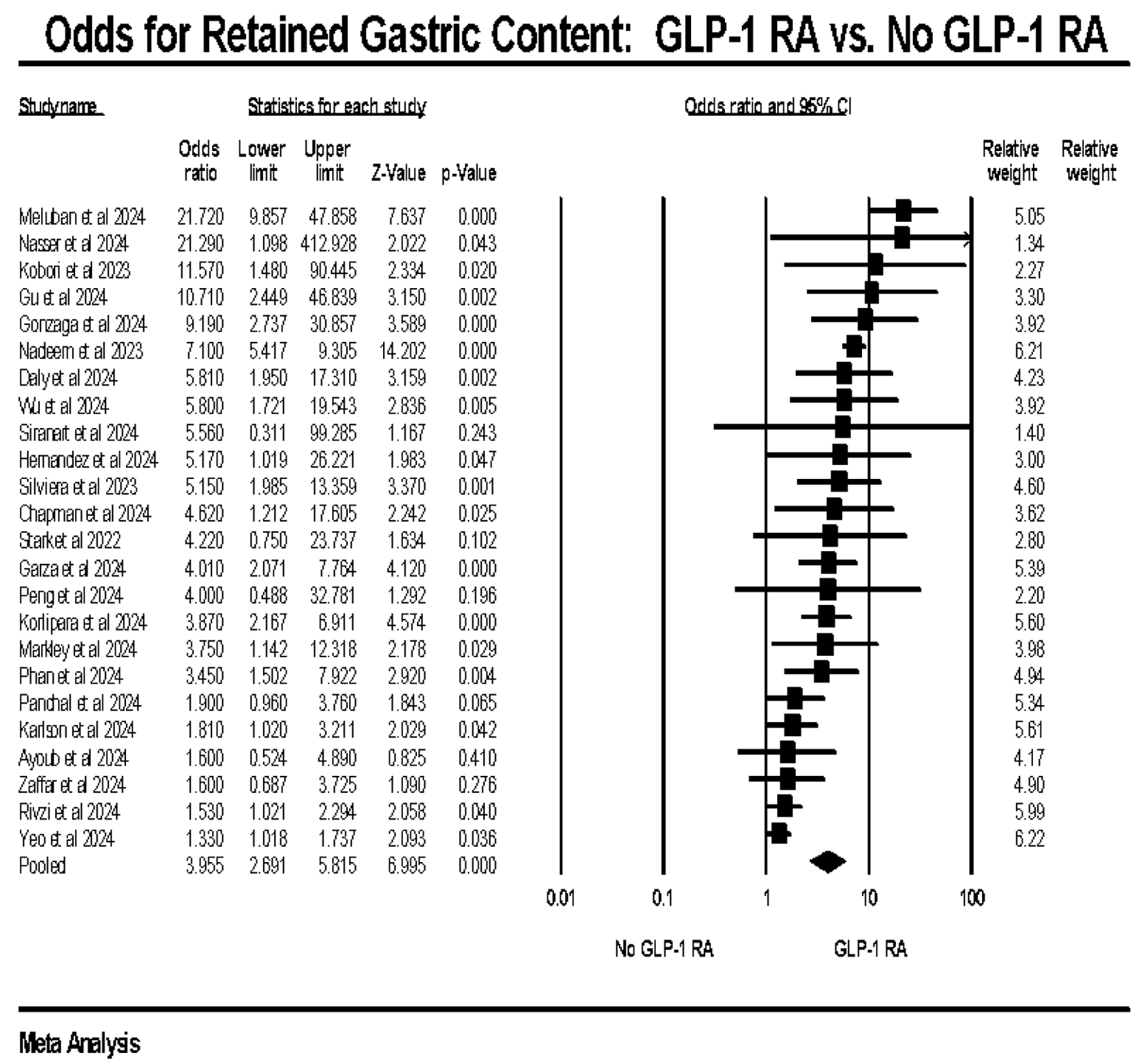 Click for large image | Figure 2. Primary outcome: odds of retained gastric content in GLP-1 RA vs. no GLP-1 RA exposure. GLP-1 RA: glucagon-like peptide-1 receptor agonist. |
Secondary outcomes
GLP-1 RA exposure was not associated with aspiration (OR: 1.06, CI: 0.466–2.439, P = 0.145, I2: 91.44%) (Fig. 3a) or increased risk of the need for repeat upper endoscopy (OR: 1.78, CI: 0.812–3.930, P = 0.145, I2: 85.32%) (Fig. 3b). GLP-1 RA use was associated with higher number of EGD cancellations (OR: 3.80, CI: 2.813–5.133, P < 0.0001, I2: 4.19%) (Fig. 3c). Aspiration events were rare across studies, generally occurring in less than 1% of procedures in both groups.
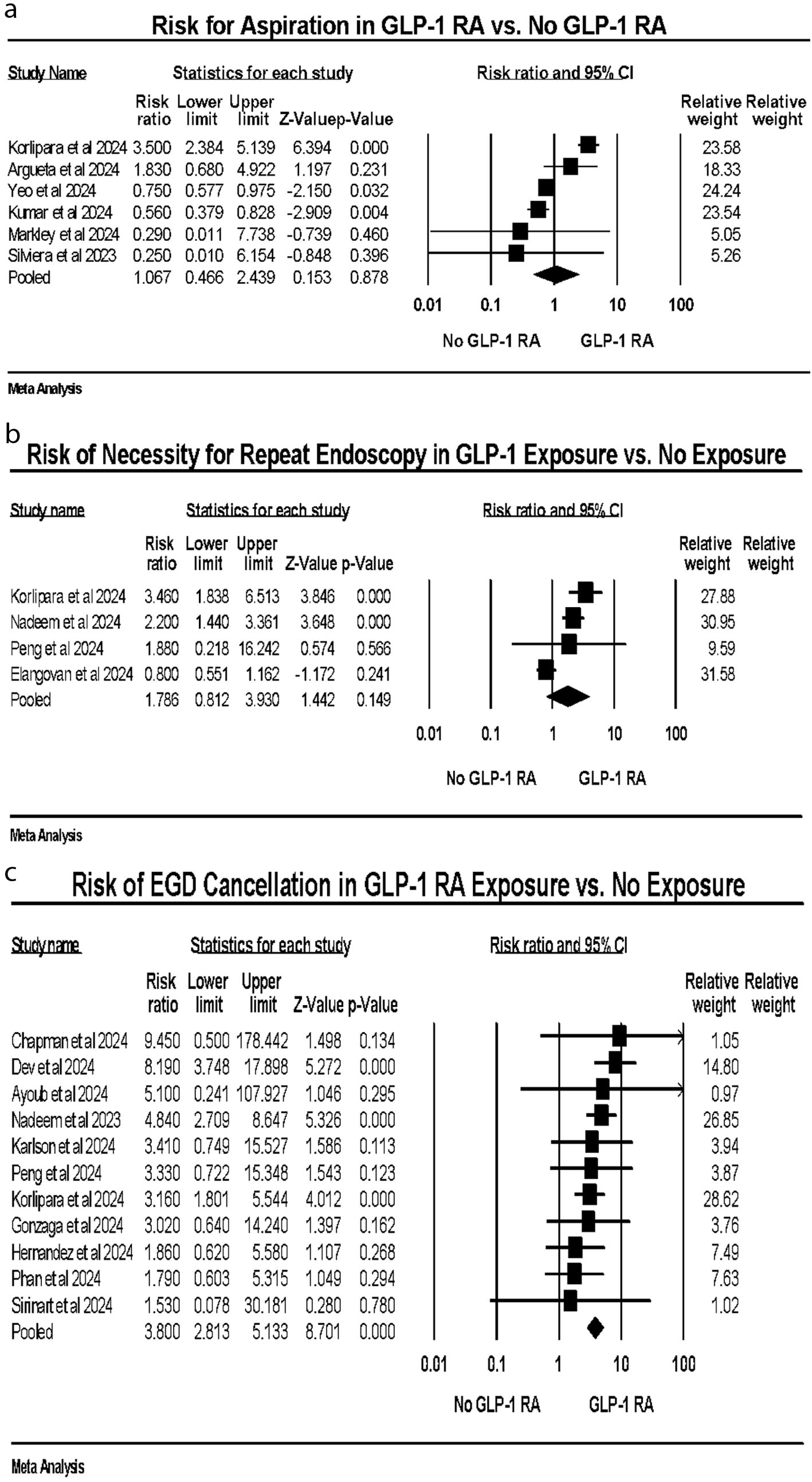 Click for large image | Figure 3. (a) Aspiration events. (b) Repeat upper endoscopy. (c) Cancellation of upper endoscopy. |
Quality assessment
Bias assessment for including observational studies using Newcastle-Ottawa scale showed a score of 6–8 for all studies (Supplementary Material 4, gr.elmerpub.com).
Validation of meta-analysis results
Our sensitivity analysis conducted by excluding abstracts from the RGC outcome revealed a pooled OR of 4.30 (CI: 1.98-9.342), n=, p < 0.0001, I2 = 78.7%, n = 8, (Supplementary Material 6a, gr.elmerpub.com). Also, sensitivity analysis performed by excluding one study sequentially did not change the effect size for RGC and cancelled procedures (Supplementary Material 6b, c, gr.elmerpub.com).
Publication bias assessment
In terms of quantitative publication bias, the Egger’s regression test for RGC and canceled upper endoscopy revealed two-tailed P-value > 0.05 indicating no significant asymmetry for observed studies. Qualitative assessment of funnel plot for RGC and cancellation of upper endoscopy revealed most effect sizes within the expected range of variation. There was some asymmetry with more studies towards the right (Supplementary Material 5, gr.elmerpub.com).
| Discussion | ▴Top |
Our primary outcome analysis revealed significantly increased odds of RGC in patients using GLP-1 RAs compared to those not using these agents. The pooled OR of 3.955 (CI: 2.691–5.815) underscores a substantial association, suggesting that GLP-1 RAs significantly contribute to the presence of RGC during endoscopic procedures. This finding is critical, as RGC can impair visibility and therefore limit the procedure, potentially leading to diagnostic inaccuracies and procedural delays. The high heterogeneity (I2 = 82.71%) observed in our analysis highlights the variability among the included studies, which may be attributed to differences in study populations, GLP-1 RA types, and study designs.
Our findings are consistent with other studies that have reported an association between GLP-1 RA use and increased gastric content during endoscopy. For instance, Kobori et al [8] found that patients on GLP-1 RAs had a higher incidence of RGC, which impacted the quality of endoscopic examinations. Similarly, Ghazanfar et al [46] observed that GLP-1 RA users were more likely to experience procedural complications due to residual gastric material. These studies reinforce the need for careful management of patients on GLP-1 RAs to mitigate risks associated with endoscopic procedures.
For secondary outcomes, our analysis did not demonstrate a statistically significant increase in aspiration events among patients receiving GLP-1 RAs compared to non-users. However, aspiration events were rare across included studies, and substantial heterogeneity was observed (I2 >90%). Therefore, the absence of a statistically significant association should be interpreted cautiously, as this analysis may be underpowered to detect small but clinically meaningful differences in aspiration risk. Further studies are needed to clarify this relationship. This result aligns with studies such as those by Yeo et al [18] and Barlowe et al [47] which also found no significant difference in aspiration events among patients using GLP-1 RAs compared to non-users. These studies indicate that while GLP-1 RAs are associated with other procedural risks, their impact on aspiration events may be minimal or influenced by other confounding factors. Further research is essential to explore these variables and develop a comprehensive understanding of aspiration risk in patients on GLP-1 RAs.
The risk of needing a repeat endoscopy did not meaningfully differ in GLP-1 RA users when compared to non-GLP-1 RA users, with a pooled OR of 1.822 (CI: 0.813–4.085); this did not reach statistical significance (P = 0.145). The increased need for repeat procedures could be related to the presence of RGC, which compromises initial endoscopy quality. This finding emphasizes the importance of optimizing patient preparation protocols to reduce the likelihood of repeat procedures and associated healthcare costs. Consistent with our findings, Stark et al [10] observed an elevated risk of repeat endoscopy in patients on GLP-1 RAs due to poor visualization conditions during the initial procedure. Furthermore, Nadeem et al [37] highlighted the economic implications of repeat endoscopies, emphasizing the need for better pre-procedure management to prevent additional healthcare expenditures. These studies reinforce the necessity for tailored patient preparation protocols for those on GLP-1 RAs.
Notably, the risk of EGD cancellation was significantly higher in patients on GLP-1 RAs, with a pooled OR of 3.966, CI: 2.870–5.563 and minimal observed heterogeneity (I2 = 4.14%). This suggests a consistent and strong association across studies, indicating that GLP-1 RA use is a substantial risk factor for EGD cancellation. This outcome is particularly concerning as it directly impacts patient safety and procedural success rates and has downstream effects on financial aspects of endoscopy, patient and physician workflow, and other relevant issues. Consistent with our findings, Mizubuti et al [48] reported a higher rate of EGD cancellations in patients on GLP-1 RAs due to inadequate gastric emptying. Similarly, Peng et al [40] found that the use of GLP-1 RAs significantly increased the likelihood of procedure cancellations, highlighting the need for improved management strategies for these patients.
This is one of the most extensive systematic reviews and meta-analyses on this topic, encompassing 28 retrospective studies with a large sample size of 212,082 patients. This comprehensive approach enhances the generalizability and robustness of our findings. Furthermore, by including both cohort studies and case-control studies, our analysis benefits from a variety of perspectives and methodologies, which adds depth and reliability to our conclusion. Finally, sensitivity analysis that excludes abstracts strengthens the validity of our findings by addressing potential biases and confirming the robustness of the primary outcome. The use of propensity-matched cohorts when available strengthens internal validity but does not eliminate residual confounding.
Our meta-analysis does have a few limitations. First, the reliance on retrospective studies introduces inherent biases, such as recall bias and selection bias, which can affect the accuracy and reliability of the results. Prospective studies are needed to validate these findings. Second, the high heterogeneity observed in some analyses (e.g., RGC and aspiration events) suggests variability in study populations, GLP-1 RA types, dosages, and study designs. This heterogeneity may limit the applicability of the pooled results to specific clinical settings. Although subgroup analyses or meta-regression could further explore sources of heterogeneity, these were not feasible due to inconsistent reporting across studies and lack of stratified data. This limits the ability to identify specific drivers of variability in the pooled estimates.
Third, the studies included in our analysis did not consistently differentiate between different GLP-1 RAs. Variations in the pharmacokinetics and pharmacodynamics of different GLP-1 RAs could influence the outcomes, and further research is needed to explore these differences. Fourth, the potential for publication bias exists, as studies with significant findings are more likely to be published. This bias could skew the results of the meta-analysis and overestimate the risks associated with GLP-1 RA use. Fifth, the quality of the studies included varies, and some studies may have methodological weaknesses that could impact the overall findings. Sixth, the absence of RCTs in our analysis limits the strength of the evidence. Last, several studies included in our analysis are only available as abstracts, and the full manuscripts are not accessible. This limitation may impact the depth of data extraction and the overall quality of evidence, as abstracts often provide limited information compared to full-text articles. Additionally, definitions of retained gastric content were not uniform across studies, which may have contributed to the observed heterogeneity and limit direct comparability of results. Also, timing of the last GLP-1 RA dose prior to endoscopy was inconsistently reported, which may influence gastric emptying and contribute to heterogeneity in outcomes. Additionally, included studies reported a mix of adjusted and unadjusted estimates; pooling these may introduce methodological variability and does not fully account for residual confounding.
From a practical standpoint, these findings have important implications for endoscopists and anesthesia providers. Given the increased likelihood of RGCs and procedure cancellation among patients receiving GLP-1 RAs, pre-procedural assessment should include routine screening for GLP-1 RA use and evaluation for symptoms suggestive of delayed gastric emptying, such as nausea, vomiting, or early satiety. At the time of scheduling, consideration may be given to procedure timing relative to GLP-1 RA dosing, particularly for long-acting agents, and adherence to evolving multi-society guidance regarding temporary withholding of these medications when clinically appropriate.
On the day of the procedure, heightened awareness of potential RGCs is warranted, even in patients who have adhered to standard fasting protocols. In selected patients, particularly those with symptoms or high-risk features, clinicians may consider delaying the procedure, extending fasting duration, or modifying anesthesia approach. However, given the limited prospective evidence, peri-procedural management should remain individualized until more definitive guidance is available.
In summary, our meta-analysis highlights several critical considerations regarding the use of GLP-1 RAs in patients undergoing upper endoscopy. The increased risk of RGC and EGD cancellation associated with GLP-1 RA use underscores the need for careful patient management and potentially modifying pre-endoscopy protocols for these patients.
Conclusions
What is already known on this topic
Given the rising prevalence of GLP-1 RA use and the critical importance of patient safety during upper endoscopic procedures, a systematic review and meta-analysis was needed to rigorously evaluate the potential impact of GLP-1 RAs on endoscopic outcomes. Our study aims to provide a clearer understanding of the risks associated with GLP-1 RA use in the context of upper endoscopy, addressing previous uncertainties and informing clinical decision-making. By synthesizing data from multiple studies, our analysis seeks to guide healthcare providers in balancing the benefits of GLP-1 RAs with potential procedural risks.
What this study adds
This study reveals that GLP-1 RAs significantly increase the odds of RGCs and procedure cancellations during upper endoscopy. However, they do not increase the risk of aspiration or the need for repeated procedures. These findings clarify the specific risks associated with GLP-1 RA use before endoscopy, guiding safer clinical practices.
How this study might affect research, practice, or policy
This study highlights the need for careful pre-endoscopy assessment of patients on GLP-1 RAs, potentially leading to revised guidelines to minimize procedure cancellations. It encourages further research into alternative management strategies to mitigate risks associated with gastric retention. Policymakers might consider these findings when updating protocols for endoscopic procedures in patients using GLP-1 RAs.
| Supplementary Material | ▴Top |
Suppl 1. Full search strategies.
Suppl 2. PRISMA guidelines.
Suppl 3. Baseline characteristics of all studies included in systematic review of GLP-1 RA vs. no GLP-1 RA on upper endoscopy.
Suppl 4. Newcastle-Ottawa scale for quality assessment.
Suppl 5. Publication bias.
Suppl 6. Sensitivity analysis.
Acknowledgments
None to declare.
Financial Disclosure
No funding was provided for the completion of this study.
Conflict of Interest
All authors have no conflict of interest to disclose.
Informed Consent
Not applicable.
Author Contributions
Zohaib Ahmed: lead author, original idea, literature review, data extraction. Omar Horani: senior author, project overseeing, manuscript review and submission process. Amna Iqbal, Ali Nawras, Yaseen Alastal, Abdallah Kobeissy, and Sami Ghazaleh: data review. Dushyant Singh Dahiya, Umair Iqbal, Syeda Faiza Arif, Connor Campbell, Hani Saad, Sarmed Mansur, Wade Lee-Smith, Mohammad W. Asif, Faisal Kamal, Aamir Syed, and Idrees Mohiuddin: manuscript review and writing.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Sheahan KH, Wahlberg EA, Gilbert MP. An overview of GLP-1 agonists and recent cardiovascular outcomes trials. Postgrad Med J. 2020;96(1133):156-161.
doi pubmed - Gorgojo-Martinez JJ, Mezquita-Raya P, Carretero-Gomez J, Castro A, Cebrian-Cuenca A, de Torres-Sanchez A, Garcia-de-Lucas MD, et al. Clinical recommendations to manage gastrointestinal adverse events in patients treated with Glp-1 receptor agonists: a multidisciplinary expert consensus. J Clin Med. 2022;12(1).
doi pubmed - Bahtiyar G, Pujals-Kury J, Sacerdote A. Cardiovascular effects of different GLP-1 receptor agonists in patients with type 2 diabetes. Curr Diab Rep. 2018;18(10):92.
doi pubmed - Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, McGowan BM, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002.
doi pubmed - Sarma S, Palcu P. Weight loss between glucagon-like peptide-1 receptor agonists and bariatric surgery in adults with obesity: a systematic review and meta-analysis. Obesity (Silver Spring). 2022;30(11):2111-2121.
doi pubmed - Arnold SV, Tang F, Cooper A, Chen H, Gomes MB, Rathmann W, Shimomura I, et al. Global use of SGLT2 inhibitors and GLP-1 receptor agonists in type 2 diabetes. Results from DISCOVER. BMC Endocr Disord. 2022;22(1):111.
doi pubmed - Mahtta D, Ramsey DJ, Lee MT, Chen L, Al Rifai M, Akeroyd JM, Vaughan EM, et al. Utilization rates of SGLT2 inhibitors and GLP-1 receptor agonists and their facility-level variation among patients with atherosclerotic cardiovascular disease and type 2 diabetes: insights from the department of veterans affairs. Diabetes Care. 2022;45(2):372-380.
doi pubmed - Kobori T, Onishi Y, Yoshida Y, Tahara T, Kikuchi T, Kubota T, Iwamoto M, et al. Association of glucagon-like peptide-1 receptor agonist treatment with gastric residue in an esophagogastroduodenoscopy. J Diabetes Investig. 2023;14(6):767-773.
doi pubmed - Silveira SQ, da Silva LM, de Campos Vieira Abib A, de Moura DTH, de Moura EGH, Santos LB, Ho AM, et al. Relationship between perioperative semaglutide use and residual gastric content: A retrospective analysis of patients undergoing elective upper endoscopy. J Clin Anesth. 2023;87:111091.
doi pubmed - Stark JE, Cole JL, Ghazarian RN, Klass MJ. Impact of Glucagon-Like Peptide-1 Receptor Agonists (GLP-1RA) on Food Content During Esophagogastroduodenoscopy (EGD). Ann Pharmacother. 2022;56(8):922-926.
doi pubmed - Patel J, Sohal A, Chaudhry H, Kalra S, Kohli I, Singh I, Dukovic D, et al. Predictors and impact of aspiration pneumonia in patients undergoing esophagogastroduodenoscopy: national inpatient sample 2016-2020. Eur J Gastroenterol Hepatol. 2024;36(3):298-305.
doi pubmed - Lin E, Gao Y, Wright A, Kim JJ. Practice patterns and outcomes of patients with retained gastric food content encountered during endoscopy. Clin Transl Gastroenterol. 2023;14(3):e00559.
doi pubmed - Lim JU, Park JJ, Youn YH, Kim S, Jeon JW, Jung SW, Shin HP, et al. Predictive factors for endoscopic visibility and strategies for pre-endoscopic prokinetics use in patients with upper gastrointestinal bleeding. Dig Dis Sci. 2015;60(4):957-965.
doi pubmed - Ushakumari DS, Sladen RN. ASA Consensus-based Guidance on Preoperative Management of Patients on Glucagon-like Peptide-1 Receptor Agonists. Anesthesiology. 2024;140(2):346-348.
doi pubmed - Singh S, Suresh Kumar VC, Aswath G. Impact of glucagon-like peptide receptor agonists on endoscopy and its preoperative management: Guidelines, challenges, and future directions. World J Gastrointest Endosc. 2024;16(6):292-296.
doi pubmed - Hashash JG, Thompson CC, Wang AY. AGA rapid clinical practice update on the management of patients taking GLP-1 receptor agonists prior to endoscopy: communication. Clin Gastroenterol Hepatol. 2024;22(4):705-707.
doi pubmed - Chapman MB, Norwood DA, Price C, Abdulhadi B, Kyanam Kabir Baig K, Ahmed AM, Peter S, et al. Effects of glucagon-like peptide-1 receptor agonists on gastric mucosal visibility and retained gastric contents during EGD. Gastrointest Endosc. 2024;100(5):923-927.
doi pubmed - Yeo YH, Gaddam S, Ng WH, Huang PC, Motility, Metabolic Pharmacoepidemiology G, Ma KS, et al. Increased risk of aspiration pneumonia associated with endoscopic procedures among patients with glucagon-like peptide 1 receptor agonist use. Gastroenterology. 2024;167(2):402-404.e403.
doi pubmed - Wu F, Smith MR, Mueller AL, Klapman SA, Everett LL, Houle T, Kuo B, et al. Association of glucagon-like peptide receptor 1 agonist therapy with the presence of gastric contents in fasting patients undergoing endoscopy under anesthesia care: a historical cohort study. Can J Anaesth. 2024;71(7):958-966.
doi pubmed - Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
doi pubmed - Scherer RW, Saldanha IJ. How should systematic reviewers handle conference abstracts? A view from the trenches. Syst Rev. 2019;8(1):264.
doi pubmed - Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557-560.
doi pubmed - Analyzing the use of glucagon-like peptide-1 agonists prior to endoscopy in bariatric patients. Surg Obes Relat Dis, 2024; 20(6):S109.
- Argueta PP, et al. GLP-1 agonist use is not associated with worse outcomes or increased mortality post endoscopy: A multicenter analysis. 2024; AB232.
- Ayoub M, et al. The impact of GLP-1 receptor agonist use prior to esophagogastroduodenoscopy. 2024; AB261.
- Essop T, Tran K, Purdy AC, Daly SC. Comparative effects of GLP-1 agonists, sleeve gastrectomy and Roux-en-Y gastric bypass on diabetes mellitus outcomes. Curr Diab Rep. 2024;24(12):273-289.
doi pubmed - Dev B, et al. Low rates of gastric food retention during endoscopy in patients on GLP-1 receptor agonists at a large academic center. 2024;AB284.
- Elangovan A, Shah R. Rates of repeat upper endoscopy in non-diabetic adults with glucagon-like peptide prescription. Gastroenterology, 2024;166(5):S207-S208.
- Garza K, et al. GLP-1 receptor agonists increase solid gastric residue rates on upper endoscopy, especially in complicated diabetic patients. 2024;AB382-AB383.
- Robalino Gonzaga E, Farooq A, Mohammed A, Chandan S, Fawwaz B, Singh G, Malik A, et al. Real-world impact of GLP-1 receptor agonists on endoscopic patient outcomes in an ambulatory setting: a retrospective study at a large tertiary center. J Clin Med. 2024;13(18).
doi pubmed - Gu GH, et al. The impact of semaglutide on retained gastric contents during endoscopy. Gastroenterology. 2024;166(5):S599-S600.
- Hernandez L, et al. GLP-1 receptor agonist effect on retained gastric contents on EGD: A quality improvement study. 2024;AB168-AB169.
- Karlson R, et al. Clinical impact of GLP-1RA on endoscopy: A retrospective cohort study in endoscopy patients. Gastroenterology. 2024;166(5):S1197.
- Korlipara H, et al. Semaglutide is an independent risk factor for retained gastric contents and recommendation for repeat upper endoscopy: A real-world cohort study. 2024;AB147.
- Markley A, et al. GLP-1 receptor agonist use predicts retained gastric contents but not increased anesthesia-related adverse events during upper endoscopy. 2024;AB1139.
- Meluban L, Alter A, Spinnell M. Increased risk of residual gastric content in patients on GLP-1 agonists undergoing EGD. Gastroenterology. 2024;166(5):S1189-S1190.
- Nadeem D, et al. Effects of glucagon-like peptide-1 receptor agonists on upper endoscopy in diabetic and non-diabetic patients. Gastrointest Endosc. 2024.
- Nasser J, et al. Colonoscopy preparation may mitigate food retention in patients using GLP-1 receptor agonists undergoing upper endoscopy. Gastroenterology. 2024;166(5):S1190-S1191.
- Panchal S, et al. Outcomes associated with GLP-1 receptor agonist use in patients undergoing outpatient upper endoscopy. Gastroenterology. 2024;166(5):S207.
- Peng FB, et al. GLP-1 receptor agonists: Impact on outpatient upper endoscopy procedure quality. Gastroenterology. 2024;166(5):S646.
- Phan J, Chang P, Issa D, Turner R, Dodge J, Westanmo A, Karna R, et al. Glucagon-like peptide receptor agonists use before endoscopy is associated with low retained gastric contents: a multicenter cross-sectional analysis. Am J Gastroenterol. 2025;120(3):554-561.
doi pubmed - Rizvi A, et al. Endoscopic gastric food retention rates for GLP-1RA users can be mitigated by pre-colonoscopy fasting protocols. 2024;AB230-AB231.
- Kumar S, VC, et al. Incidence of aspiration pneumonitis and emergent intubation in patients on GLP-1 inhibitors undergoing upper endoscopy and colonoscopy. 2024;AB150-AB151.
- Siranart N, Thompson C, Jirapinyo P. GLP-1 receptor agonist and upper endoscopy: A real-world experience. 2024;AB196.
- Zaffar D, et al. Association between perioperative GLP-1 agonist use and postoperative complications in patients undergoing upper endoscopy. Gastroenterology. 2024;166(5):S653-S654.
- Ghazanfar H, Javed N, Qasim A, Sosa F, Altaf F, Khan S, Mahasamudram J, et al. Is it necessary to stop glucagon-like peptide-1 receptor agonists prior to endoscopic procedure? A retrospective study. World J Gastroenterol. 2024;30(26):3221-3228.
doi pubmed - Barlowe TS, Anderson C, Sandler RS, Subramaniam D, Muratore A, Buse JB, Gouker LN, et al. Glucagon-like peptide-1 receptor agonists do not increase aspiration during upper endoscopy in patients with diabetes. Clin Gastroenterol Hepatol. 2025;23(5):739-747.
doi pubmed - Li XY, Jin Y, Feng XY, Wang RC, Chen JP, Lu B. Perioperative management of patients on GLP-1 receptor agonists: risks, recommendations, and future directions-A narrative review. J Clin Anesth. 2025;104:111871.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Gastroenterology Research is published by Elmer Press Inc.