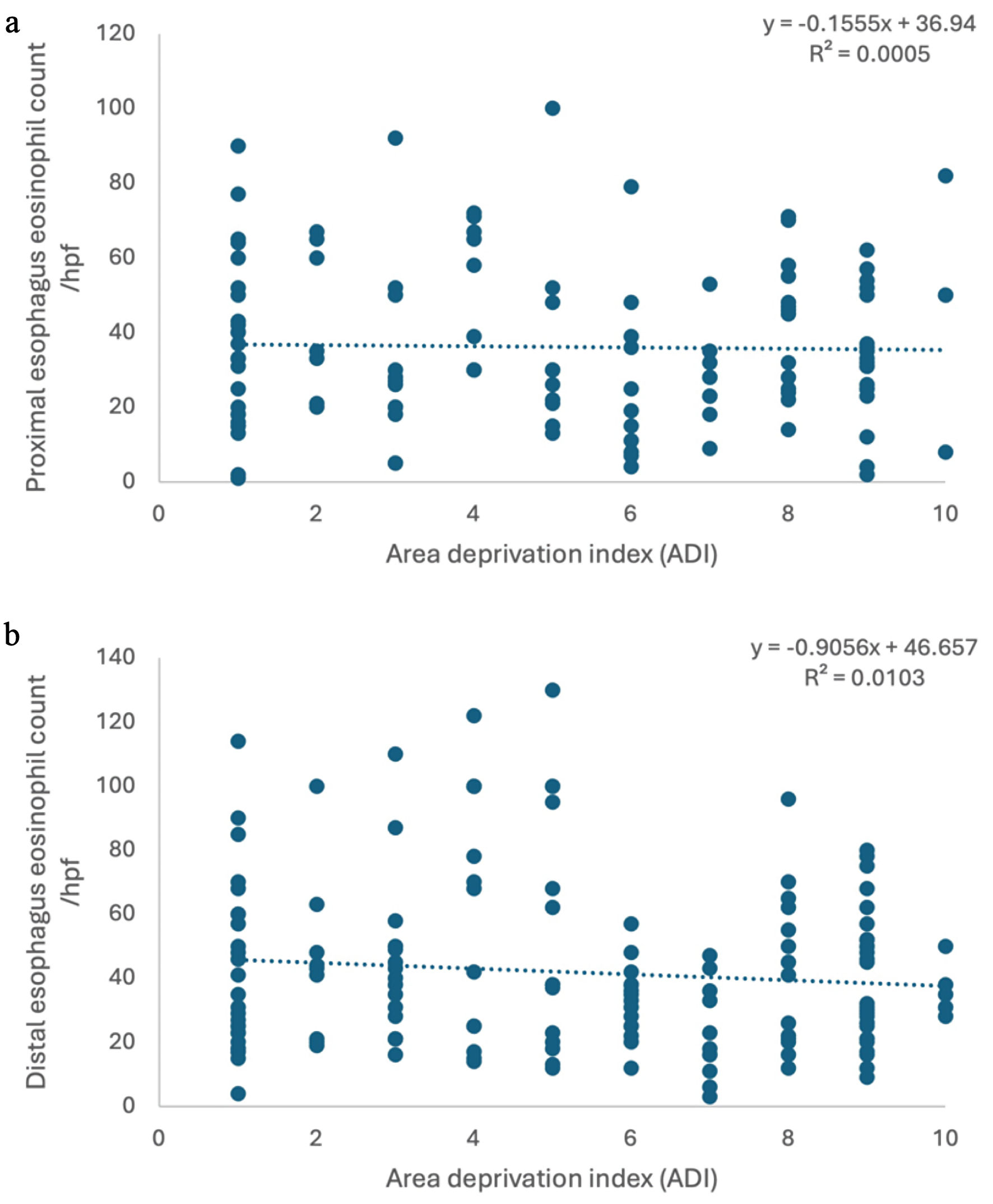
↓ Figure 1. No correlation was found between ADI and eosinophil count in proximal esophagus (a) and distal esophagus (b). ADI: Area Deprivation Index.
| Gastroenterology Research, ISSN 1918-2805 print, 1918-2813 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Gastroenterol Res and Elmer Press Inc |
| Journal website https://gr.elmerpub.com |
Short Communication
Volume 19, Number 2, April 2026, pages 110-117
Socioeconomic Deprivation Does Not Influence Disease Severity or Access to Dupilumab in Eosinophilic Esophagitis
Figures

Tables
| ADI: Area Deprivation Index; SD: standard deviation. | |
| Age at diagnosis (1–18 years), mean ± SD | 8.6 ± 5.05 |
| Sex | |
| Male | 68% |
| Female | 32% |
| Race | |
| White | 73% |
| Black | 22% |
| Hispanic | 3% |
| ADI distribution | |
| 1–3 | 42% |
| 4–7 | 26% |
| 8–10 | 32% |
| SD: standard deviation; HPF: high power field; PPI: proton pump inhibitor; STC: swallowed topical corticosteroid. | |
| Age at diagnosis (1–18 years), mean ± SD | 8.56 ± 5.13 |
| Sex, n (%) | |
| Male | 89 (69%) |
| Female | 40 (31%) |
| Race | |
| White | 74% |
| Black | 22% |
| Hispanic | 2% |
| Baseline eosinophil count/HPF | |
| Distal esophagus | 41.7 ± 2.14 |
| Proximal esophagus | 35.4 ± 3.79 |
| Specific treatment received, n (%) | |
| Dietary elimination | 2 (2%) |
| PPI | 38 (29%) |
| STC | 59 (46%) |
| PPI + STC | 30 (23%) |
| SD: standard deviation; PPI: proton pump inhibitor; STC: swallowed topical corticosteroid. | |
| Age at diagnosis (1–17 years), mean ± SD | 8.91 ± 4.47 |
| Sex, n (%) | |
| Male | 10 (63%) |
| Female | 6 (37%) |
| Race | |
| White | 69% |
| Black | 25% |
| Hispanic | 6% |
| Comorbidities | |
| Atopic condition(s) | 75% |
| None | 25% |
| Symptom severity | |
| Food impaction | 13% |
| Prior esophageal dilation | 6% |
| Treatment(s) prior to dupilumab | |
| PPI and STC | 82% |
| STC only | 18% |
| Number of failed treatment(s) | |
| Two treatments | 100% |
| Duration of previous treatment(s) | 3–12 months |
| Reason for discontinuation | |
| Inadequate response | 62% |
| Intolerance | 25% |
| Non-compliance | 13% |