Evaluating Glucagon-Like Peptide-1 Receptor Agonist Safety Before Upper Endoscopy: A Systematic Review and Meta-Analysis
DOI:
https://doi.org/10.14740/gr2108Keywords:
Glucagon-like peptide-1 receptor agonists, Retained gastric contents, Upper endoscopy, EGDAbstract
Background: The use of glucagon-like peptide-1 receptor agonists (GLP-1 RAs) has remarkably increased in the past few years as they have demonstrated significant reduction in A1c and major cardiovascular outcomes and have led to weight loss with low risk of hypoglycemia. GLP-1 RA use has raised concerns about potential associations with increased gastric retention and adverse outcomes in upper endoscopy procedures.
Methods: We conducted a comprehensive search across multiple databases to identify comparative studies evaluating the impact of GLP-1 RA exposure versus non-exposure on upper endoscopy outcomes. Studies comparing GLP-1 RAs to other medications were excluded. The primary outcome was the presence of retained gastric contents, with secondary outcomes including aspiration risk and the need for repeated esophagogastroduodenoscopy (EGD).
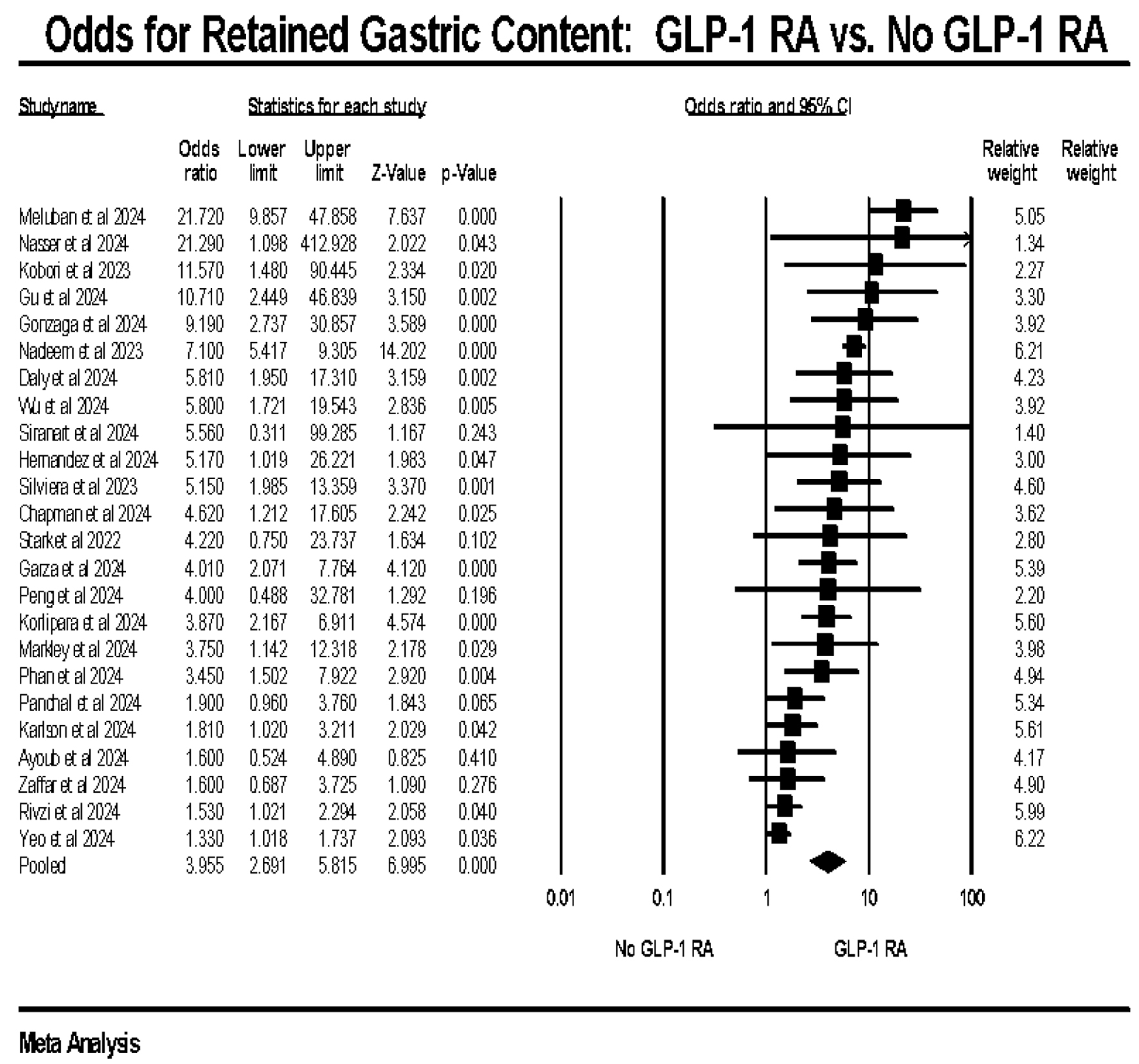
Results: Our systematic review included 28 studies, comprising data from 212,082 individuals collected between 2015 and 2023. The meta-analysis revealed that GLP-1 RA use was associated with approximately four times higher odds of retained gastric contents compared to non-exposure (odds ratio (OR): 3.95, confidence interval (CI): 2.691–5.815, P < 0.0001, I2: 82.71%). There were no increased odds of aspiration (OR: 1.09, CI: 0.459–2.600, P = 0.145, I2: 91.44%) or repeated upper endoscopies (OR: 1.82, CI: 0.813–4.085, P = 0.145, I2: 85.32%). However, GLP-1 RA use was significantly associated with increased odds of procedure cancellation (OR: 3.96, CI: 2.870–5.563, P < 0.0001, I2: 4.19%).
Conclusion: While GLP-1 RAs do not increase the risk of aspiration or necessitate repeated procedures, they are linked to a higher incidence of retained gastric contents and procedure cancellations. Further randomized controlled trials are necessary to better understand the effects of GLP-1 RAs on upper endoscopy outcomes.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









